Azurity Pharmaceuticals, Inc. Issues Voluntary Nationwide Recall of One Lot of Firvanq® (vancomycin hydrochloride for oral solution), Vancomycin 50 mg/mL Kit, Due to a Mix-Up of the Diluent Included in the Kit
Company Contact:
Koral Couch
781-935-8141 x119
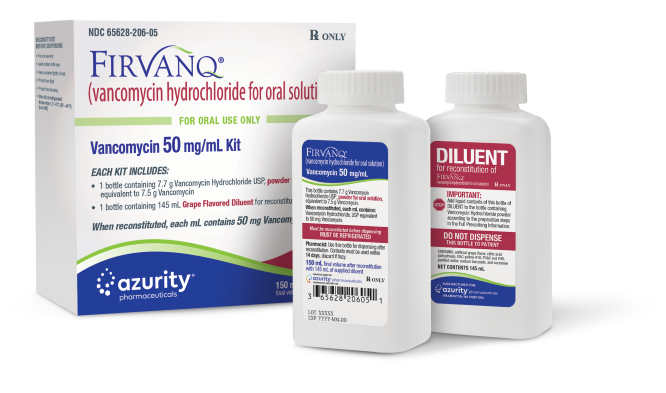
FOR IMMEDIATE RELEASE – September 7, 2021 – Wilmington, MA, Azurity Pharmaceuticals, Inc. (“Azurity”) is voluntarily recalling one lot of Firvanq® (vancomycin hydrochloride for oral solution), Vancomycin 50 mg/mL Kit (“Firvanq®”) to the consumer level, as some products in the affected lot have been found to incorrectly contain a First Omeprazole (FIRST-PPI) diluent instead of the Firvanq diluent bottle.
Risk Statement: Vancomycin may not be completely solubilized in the FIRST-PPI diluent which could lead to doses above or below those recommended in the label. There is reasonable probability that the administration of inappropriate doses of oral vancomycin may lead to persistent diarrhea associated with dehydration and electrolyte abnormalities, recurrence of Clostridium difficile (C. difficile) infection, its progression to severe colitis, colon perforation requiring colectomy, and potentially death. Especially, the elderly and immunocompromised patients are vulnerable to the complications of C. difficile infection. To date, Azurity has not received any reports of adverse events related to this recall.
Firvanq® is indicated for use in adults and pediatric patients less than 18 years of age for the treatment of: Clostridium difficile-associated diarrhea and Enterocolitis caused by Staphylococcus aureus (including methicillin-resistant strains). It is packaged as a kit consisting of a bottle with Vancomycin Hydrochloride, USP powder, a bottle of Grape-Flavored diluent, and Full Prescribing Information. The product NDC is 65628-206-05 and the UPC code is 3 65628 206005 1. A total of 2,751 kits of the affected Firvanq®, lot number 21035, with an expiration date of 2022-07-31 were distributed. The product can be identified by its brand name Firvanq®. Product was distributed nationwide through wholesale distributors.
Azurity is notifying its distributors and customers by direct notifications to distributors and via this press release. Azurity is arranging for return and replacement of only recalled products. Consumers, distributors, and retailers that are in possession of Firvanq® from the affected lot should immediately stop using it and return it to the place of purchase. Distributors are asked to place any affected product under quarantine and return it promptly to Azurity.
Consumers with questions regarding this recall may contact Koral Couch, Senior Manager, Customer Service by phone: 781 935-8141 x 119; fax: 781 935-8395 or email: kcouch@azurity.com. Azurity business hours are Monday through Friday from 8:30 am to 5 pm Eastern Time. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- -Complete and submit the report Online: https://www.fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program
- -Regular Mail or Fax: Download form https://www.fda.gov/safety/medical-product-safety-information/medwatch-forms-fda-safety-reporting or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.

